Making Proteins from Peptides: Uncovering the Role of Posttranslational Modifications in Human Disease
Michelson Center for Convergent Biosciences
University of Southern California, Los Angeles, CA USA
O-GlcNAc is a dynamic form of intracellular glycosylation found throughout the nucleus, cytosol, and mitochondria. This posttranslational modification is essential for development in mammals and changes in O-GlcNAc levels are associated with cancer, diabetes, and neurodegenerative diseases. Proteomics experiments have identified thousands of O-GlcNAc modified proteins, but the consequences of the vast majority of these modification events (if any) are totally unknown. To address this lack of understanding, my lab uses protein ligation to synthesize site-specifically O-GlcNAc modified proteins for subsequent biological experiments.
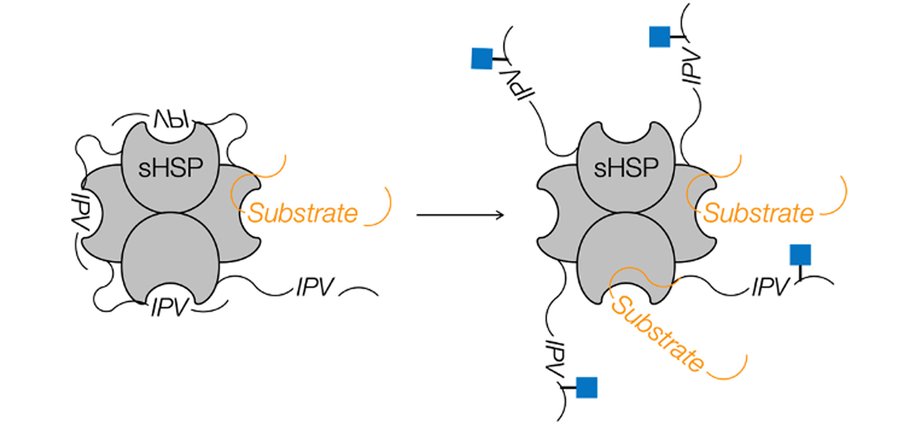
Small heat shock proteins (sHSPs) are small ATP-independent chaperones that bind to largely hydrophobic segments of proteins, preventing their further aggregation. A subset of sHSPs were some of the first O-GlcNAc modified proteins to be identified, but the effect of this modification was totally unexplored. Using synthetic protein chemistry, we build seven different glycoforms of three sHSPs and found that O-GlcNAc increases the chaperone activity of all these proteins against amyloid aggregating proteins found in neurodegenerative diseases. We also identified the molecular mechanism, where O-GlcNAc disrupts an intra-protein inhibitory interaction within the sHSPs. Finally, we find that this same mechanism increases other activities of HSP27 including protein refolding in collaboration with HSP70 and inhibition of apoptosis.
References
- Balana AT, Levine PM, Craven TW, Mukerjee S, Pedowitz NJ, Moon SP, Takahashi TT, Becker CFW, Baker D, Pratt MR. O-GlcNAcylation of small heat shock proteins enhances their anti-amyloid chaperone activity. Nat Chem 13, 441-450 (2021).
- Wang B, Moon SP, Cutolo G, Javed A, Ahn BS, Ryu AH, Pratt MR. HSP27 inhibitory activity against caspase-3 cleavage and activation by caspase-9 is enhanced by chaperone O-GlcNAc modification in vitro. ACS Chem Biol 18, 1698-1704 (2023).
- Javed A, Johnson OT, Balana AT, Volk RF, Langen A, Ahn BS, Zaro BW, Gestwicki JE, Pratt MR. O-GlcNAc modification of HSP27 alters its protein interactions and promotes refolding of proteins through the BAG3/HSP70 co-chaperone. Protein Sci 33, e5173 (2024).