Glycopeptide Drugs from Endogenous Peptide Neurotransmitters and Hormones
The University of Arizona, Tucson AZ 85721
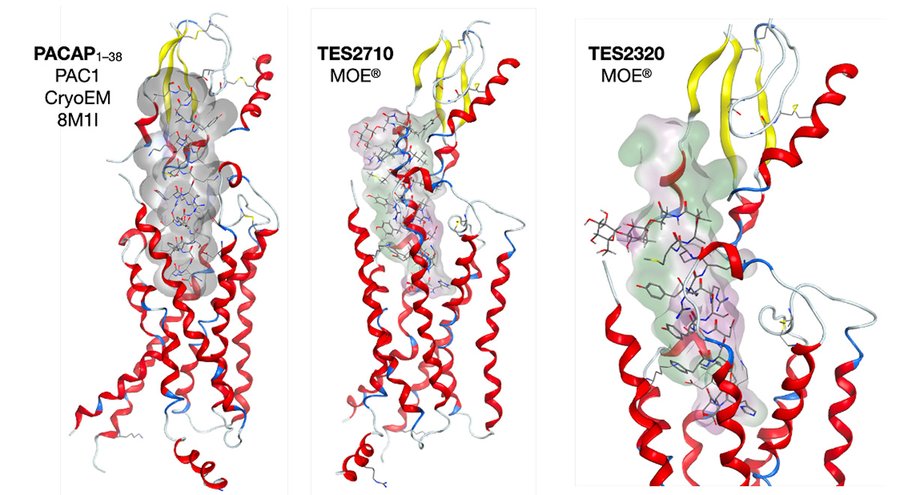
Our studies with glycopeptide drugs suggest that two conformational macrostates exist — 1) A large water-soluble ensemble of structures (random coil state). 2) A much smaller amphipathic ensemble that is membrane bound. It seems that most if not all endogenous neuropeptides possess amphipathic character, which constrains them to the membrane compartment. Pioneering studies with enkephalins and endorphin/dynorphin analogues done in collaboration with Victor Hruby suggested that the modulation of membrane affinity by glycosylation produces “biousian glycopeptides” that are systemically available and can quickly cross the BBB.
Plasma Waveguide Resonance (PWR) experiments performed in the Cai Laboratory with truncated glycosides based on Pituitary Adenylate Cyclase Activating Peptide (PACAP) as well as their unglycosylated peptide counterparts demonstrate low nM affinity for phosphatidyl choline (PC) bilayers (15.8–0.110 nM Kd’s), and sub-nM binding to the PAC1 GPCR receptor with Kd’s ranging from 130 fM to 12.1 pM. These extremely high affinities for the canonical PACAP receptors provide a compelling reason why the PAC1•PACAP interaction was conserved through 540 million years of evolution, with most of the evolution occurring on the Class B GPCR receptors and not with the PACAP ligand.
References
- Bernard K, Dickson D, Anglin BL, Leandro Heien M, Polt R, Morrison HW, Falk T. PACAP glycosides promote cell outgrowth in vitro and reduce infarct size after stroke in a preclinical model. Neuroscience Letters 836, 137883 (2024).
- Apostol CR, Bernard K, et al. Design and Synthesis of Brain Penetrant Glycopeptide Analogues of PACAP with Neuroprotective Potential for Traumatic Brain Injury and Parkinsonism. Frontiers in Drug Discovery #818003 (2022).