Mimicking Alpha-Helices for Inhibiting Protein-Protein Interactions
University of Texas at Dallas, Richardson TX 75080
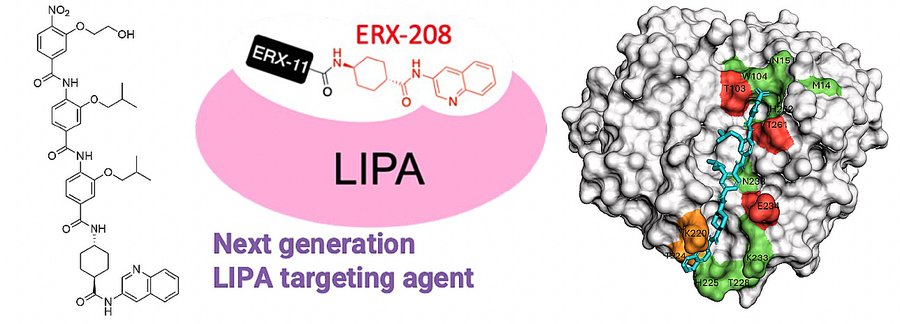
We have designed an oligo-benzamide scaffold as a rigid template to replicate protein helices. It presents side chains of the amino acid residues found at the i, i+4, and i+7 positions in a helix in addition to two substituents at its N- and C-termini for achieving higher affinity, selectivity, and improved physicochemical properties. It has demonstrated outstanding mimicry of LXXLL motifs that is critical to facilitate protein-protein interactions between nuclear receptors and their coactivators. Bis-benzamide D2 was found to disrupt androgen receptor and its coactivators effectively in prostate cancer models, whereas tris-benzamide ERX-11 showed potent inhibition of estrogen receptor in breast cancer models. Recently, our efforts to further improve these leads identified a new therapeutic target, lysosomal acid lipase A (LIPA) for treating hard-to-kill cancers like triple-negative breast cancer. Tris-benzamide-based ERX-41 was found to induce endoplasmic reticulum (ER) stress, resulting in cell death. Mechanistically, ERX-41 binding to LIPA decreases expression of multiple ER-resident proteins involved in protein folding. This targeted vulnerability has a large therapeutic window with no adverse effects either on normal mammary epithelial cells or in mice. It is also found to be metabolically stable and orally available. Iterative structure–activity relationship–guided studies to enhance activity in ovarian cancer cells identified a more potent ERX-41 analog, ERX-208. It binds to LIPA over a broader interaction surface, examined through in silico molecular docking simulation and site-directed mutagenesis.
References
- Vadlamudi RK, Viswanadhapalli S, et al. Therapeutic optimization of LIPA targeting to induce endoplasmic reticulum stress and cell death in Ovarian Cancer. Oncogene 45, 790-804 (2026).
- Lee TK, Kassees K, Chen CY, Viswanadhapalli S, Parra K, Vadlamudi RK, Ahn JM. Structure-Activity Relationship Study of Tris-Benzamides as Estrogen Receptor Coregulator Binding Modulators. ACS Pharmacol. Transl. Sci. 7, 2023-2043 (2024).
- Liu X, Viswanadhapalli S, et al. Identification of Lysosomal Acid Lipase A as a novel molecular vulnerability in triple negative breast cancer. Nature Cancer 3, 866-884 (2022).
- Raj GV, Sareddy GR, et al. Estrogen receptor coregulator binding modulators (ERXs) effectively target estrogen receptor positive human breast cancers. eLife 6, e26857 (2017).